Capricor Therapeutics Inc. (NASDAQ: CAPR) is a clinical-stage biotechnology company focused on the discovery, development, and commercialization of first-in-class biological therapeutics for the treatment of rare disorders. Capricor’s lead candidate, CAP-1002, is an “off-the-shelf” cardiac cell therapy that is currently in clinical development for the treatment of Duchenne muscular dystrophy.
The company recently announced that the American Heart Association (AHA) Scientific Sessions 2017 has accepted for presentation at a late-breaking session the 12-month results from the company’s HOPE-1 clinical trial of its lead investigational product, CAP-1002, in boys and young men in advanced stages of Duchenne muscular dystrophy.
Capricor previously reported that meaningful improvements in cardiac and skeletal muscle function were observed at a pre-specified six-month analysis of the HOPE-1 Trial. CAP-1002 is the company’s lead investigational product and consists of allogeneic cardiosphere-derived cells, which improved muscle function and increased new muscle cell generation in preclinical models of Duchenne muscular dystrophy.
CAPR has Plan to Initiate HOPE-2 Clinical Trial of CAP-1002 in 1Q18
- Randomized, double-blind, placebo-controlled
- Peripheral intravenous delivery – supported by preclinical studies
- Repeat-dose design – potential to achieve sustained benefit
- Primary efficacy endpoint to be based on the PUL test at six months
- FDA willing to accept PUL as an efficacy endpoint for registration
- Type B meeting held following six-month HOPE data
This is an inflection point for Capricor, as it is about to move into a phase II trial for the CAP-1002 as mentioned above. We expect that action will continue to drive Capricor higher during the upcoming periods as the pivotal trial initiates and as markets factors in a successful outcome. Several equities research analysts recently issued favorable reports on CAPR shares. On average, the consensus target is $8.30 over the medium term.
About the company: Capricor Therapeutics, Inc. (NASDAQ: CAPR) is a clinical-stage biotechnology company focused on the discovery, development, and commercialization of first-in-class biological therapeutics for the treatment of rare disorders.
Capricor’s lead candidate, CAP-1002, is an “off-the-shelf” cell therapy that is currently in clinical development for the treatment of Duchenne muscular dystrophy. Capricor has also established itself as one of the leading companies investigating the field of extracellular vesicles and is exploring the potential of CAP-2003, a cell-free, exosome-based candidate, treating a variety of disorders.
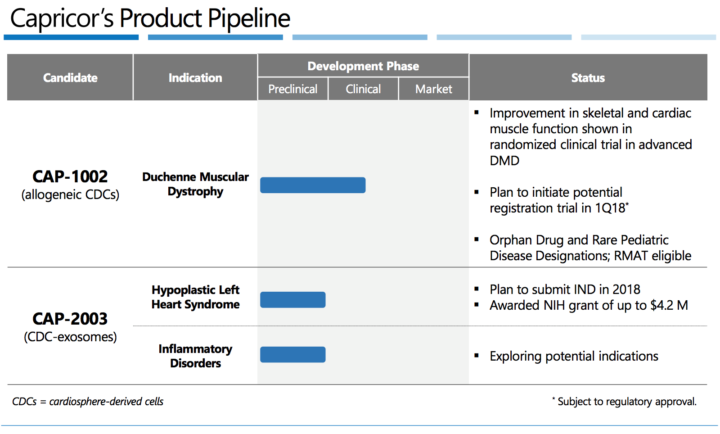
CAP – 1002: Capricor’s lead candidate, CAP-1002, is a cell therapy that is currently in clinical development for the treatment of Duchenne muscular dystrophy (DMD). CAP-1002 is an allogeneic product, meaning that it is manufactured from donor heart tissue and then stored until needed for use.
Present Trials:
Key unique factors:
- Pipeline focused on rare pediatric disorders for which current options are inadequate
- Clinical proof-of-concept demonstrated in lead indication
- Potential registration trial planned to initiate in 1Q18
- $1B+ U.S. sales opportunity
- Capricor holds worldwide IP rights
- Scalable, cost-efficient manufacturing process in development
- Disruptive technology platform that may address several challenging diseases
Recent and Upcoming Milestones
CAP-1002 in Duchenne Muscular Dystrophy
- April 2017: Reported positive top-line six-month results of HOPE trial
- July 2017: Granted Rare Paediatric Disease Designation
- July 2017: Announced results of FDA meeting
- September 2017: Announced Craig McDonald, M.D. to lead HOPE-2 clinical trial
- October 2017: Present six-month HOPE results at World Muscle Society Congress
- 4Q 2017: Submit IND for i.v. CAP-1002 in DMD with request for RMAT Designation
- 1Q 2018: Plan to initiate HOPE-2 Trial of repeat-dose, intravenous CAP-1002*
CAP-2003 2018:
- Expect to submit IND for hypoplastic left heart syndrome (HLHS)
2nd Quarter 2017 Financial Results:
- The Company reported a net loss of approximately $3.5 million, or $0.16 per share, for the second quarter of 2017, compared to a net loss of roughly $4.7 million, or $0.26 per share, for the second quarter of 2016.
- As of June 30, 2017, the Company’s cash, cash equivalents, and marketable securities totaled approximately $12.3 million compared to roughly $16.2 million on December 31, 2016. Capricor believes that its current financial resources should be sufficient to fund its operations and meet its financial obligations through the second quarter of 2018 based on the Company’s current projections.
- Upcoming Results: CAPR will release their next quarterly earnings announcement on Wednesday, November 8th, 2017.
Key risk factors and potential stock drivers:
- There is the potential upside catalyst for the company and, if news hits press favorably, the stock of the company will be on a growth trajectory. Even if it misses, the subsequent dip in share price will serve as an option to make a position for the eventual upside run.
- Notwithstanding promising data and partnerships, ONCS does not have a product yet approved by FDA. There is a risk that their potential drugs might fail during the clinical trials.
- Given the current burn rate, the company would need incremental working capital. Therefore, we might see a near-term raise and subsequent dilution.
- The company may experience financial, regulatory, or operational difficulties, which may impair its ability to commercialize their drug products.
Stock Chart:
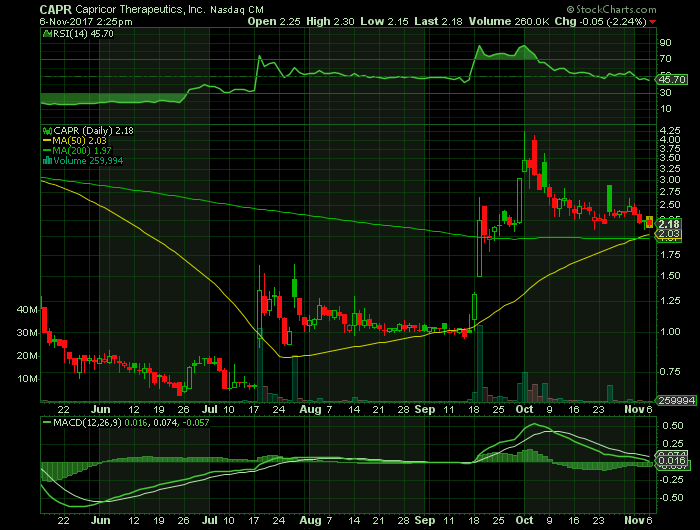
On Monday, November 6th, 2017, CAPR is trading at $2.18/share, with an above average volume of 2.32 million shares exchanging hands. Market capitalization is $56.66 million. The current RSI is 45.70
In the past 52 weeks, shares of CAPR have traded as low as $0.63 and as high as $4.25
At $2.18, shares of CAPR are trading above its 50-day moving average (MA) at $2.04 and above its 200-day MA at $1.97 as well.
The present support and resistance levels for the stock are at $2.14 & $2.34 respectively.
Welcome to Traders News Source
Our track record speaks for itself…
Traders News Source recent profiles and track record, 487% in verifiable potential gains for our members on 3 small cap alerts alone! These are just three examples from over two dozen winners this year.
January 31st, 2017 (NASDAQ: HIMX) opened at $5.10/share and hit a high of $9.68/share March 24th, 2017 for gains of 89% within 60 days- http://finance.yahoo.com/news/himax-technologies-review-4q-2016-130000319.html
May 23rd, 2016- (NYSE: XXII) opened at $.87/share hit a high of $3.03/share so far our member potential gains- 248% – http://mailchi.mp/tradersnewssource/updates-5-of-our-profiles-for-212-400-and-whats-coming-next?e=[UNIQID]
October 31st, 2017 (NASDAQ: PYDS) Although we have been covering this security for over a year, our recent coverage October 31st, 2017 opened at $1.45/share hit $4.10 within three days for gains of over 150%- http://mailchi.mp/tradersnewssource/update-pyds-back-in-the-value-zone-with-news-out?e=[UNIQID]
So, if you’ve been on the fence, perhaps it’s time to start doing some research and verify our numbers for yourself. We are constantly raising the bar and separate ourselves from the rest of the small-cap newsletters as the best in business.
We know with a large following comes a large responsibility as we have everyone from institutional investors to the beginner following our profiled securities in our newsletter. This is something we take very seriously always seeking small cap growth companies that have both near and long-term potential for our members.
Big Opportunities Trading Small Cap Stocks
***Get our small cap profiles, special situation and watch alerts in real time. We are now offering our VIP – SMS/text alert service for free, simply text the word “Traders” to the phone number “25827” from your cell phone***
Disclaimer
Traders News Source is a wholly owned subsidiary of Traders News Source LLC, herein referred to as TNS LLC.
Traders News Source has not been compensated for this report by anyone and the opinions if any are that of the author Vikas Agrawal, CFA. Author’s Disclosure: I have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours. I, wrote this article myself, and it expresses my own opinions. I have no business relationship with any company whose stock is mentioned in the article.
This web site, published by TNS LLC, and is an investment newsletter that is built on the premise of assisting individual investors in learning about investing. Our goal as publishers of financial information is to provide research and analysis of investments to our subscribers. TNS LLC does not give buy or sell recommendations. We do purchase distribution rights from analyst, financial writers and bloggers for a fee that may be licensed to issue price targets and recommendations. Furthermore, we encourage you to speak to a licensed professional prior to making an investment in any type of publicly traded security.
We do sell advertising to other companies including brokerage firms, web sites, publicly traded issuers, investor relations firms, and investment publications, among others. TNS LLC makes no warranty as to the policies of these organizations, and in no way endorses their offers, services, or the content of their advertisements.
When an advertiser is a publicly traded company or a third party acting on behalf of a public company, we fully disclose all compensation in the email advertisement. Such disclosure is included in a disclosure statement in each of the advertisements sent via email.
17B Disclosure
Our reports/releases are a commercial advertisement and are for general information purposes ONLY. We are engaged in the business of marketing and advertising companies for monetary compensation. Never invest in any stock featured on our site or emails unless you can afford to lose your entire investment. The disclaimer is to be read and fully understood before using our services, joining our site or our email/blog list as well as any social networking platforms we may use.
PLEASE NOTE WELL: TNS LLC and its employees are not a Registered Investment Advisor, Broker Dealer or a member of any association for other research providers in any jurisdiction whatsoever.
Release of Liability: Through use of this website viewing or using you agree to hold TNS LLC, its operator’s owners and employees harmless and to completely release them from any and all liability due to any and all loss (monetary or otherwise), damage (monetary or otherwise), or injury (monetary or otherwise) that you may incur. The information contained herein is based on sources which we believe to be reliable but is not guaranteed by us as being accurate and does not purport to be a complete statement or summary of the available data. TNS LLC encourages readers and investors to supplement the information in these reports with independent research and other professional advice. All information on featured companies is provided by the companies profiled, or is available from public sources and TNS LLC makes no representations, warranties or guarantees as to the accuracy or completeness of the disclosure by the profiled companies. None of the materials or advertisements herein constitute offers or solicitations to purchase or sell securities of the companies profiled herein and any decision to invest in any such company or other financial decisions should not be made based upon the information provide herein. Instead TNS LLC strongly urges you conduct a complete and independent investigation of the respective companies and consideration of all pertinent risks. Readers are advised to review SEC periodic reports: Forms 10-Q, 10K, Form 8-K, insider reports, Forms 3, 4, 5 Schedule 13D.
TNS LLC is compliant with the Can Spam Act of 2003. TNS LLC does not offer such advice or analysis, and TNS LLC further urges you to consult your own independent tax, business, financial and investment advisors. Investing in micro-cap and growth securities is highly speculative and carries an extremely high degree of risk. It is possible that an investor’s investment may be lost or impaired due to the speculative nature of the companies profiled.
The Private Securities Litigation Reform Act of 1995 provides investors a ‘safe harbor’ in regard to forward-looking statements. Any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, goals, assumptions or future events or performance are not statements of historical fact may be “forward looking statements”. Forward looking statements are based on expectations, estimates, and projections at the time the statements are made that involve a number of risks and uncertainties which could cause actual results or events to differ materially from those presently anticipated. Forward looking statements in this action may be identified through use of words such as “projects”, “foresee”, “expects”, “will”, “anticipates”, “estimates”, “believes”, “understands”, or that by statements indicating certain actions & quote; “may”, “could”, or “might” occur.
Understand there is no guarantee past performance will be indicative of future results. In preparing this publication, TNS LLC has relied upon information supplied by its customers, publicly available information and press releases which it believes to be reliable; however, such reliability cannot be guaranteed. Investors should not rely on the information contained in this website. Rather, investors should use the information contained in this website as a starting point for doing additional independent research on the featured companies. The advertisements in this website are believed to be reliable, however, TNS LLC and its owners, affiliates, subsidiaries, officers, directors, representatives and agents disclaim any liability as to the completeness or accuracy of the information contained in any advertisement and for any omissions of materials facts from such advertisement. TNS LLC is not responsible for any claims made by the companies advertised herein, nor is TNS LLC responsible for any other promotional firm, its program or its structure.

