Company Overview
Dextera Surgical Inc. (NASDAQ: DXTR) designs and manufactures products for the minimally invasive surgery market. The company’s lead product is the MicroCutter 5/80, a proprietary endoscopic stapler used in thoracic, pediatric, bariatric, colorectal, and other surgeries. The company also generates license revenue from its technology, and is co-developing a MicroCutter system with Intuitive Surgical for the da Vinci system.
Prior to 2009, Dextera focused on the design, manufacture, and marketing of proprietary automated anastomotic systems for coronary bypass surgeries. In June 2016, the company changed its name from Cardica to Dextera Surgical to reflect its shifting business model. Dextera is headquartered in Redwood City, California.
Products
The MicroCutter 5/80 is a cartridge-based stapling device with a five-millimeter shaft diameter, 80 degrees of articulation, and a 30-millimeter staple line cleared for certain specified uses in the United States, and a broader range of applications in the European Union.

Source: Company Presentation
The company’s device has several advantages over existing products. First and foremost is the shaft diameter, which at five millimeters is less than half the size of other endoscopic staplers. The MicroCutter’s 80-degree articulation (versus 45 degrees for competing devices) also provides better access to the surgical site, and the reduced anvil size minimizes high risk dissections. Dextera has received FDA 510(k) clearance to use the MicroCutter in thin tissue (white reload cartridges) and medium tissue (blue reload cartridges) for minimally invasive urologic, thoracic, and pediatric surgical procedures.
For the nine months ended March 31, 2017, the company generated revenue of $0.9 million related to the sale of MicroCutter products.
C-Port Distal Anastomosis Systems (C-Port Systems) are used for distal anastomosis, which is the connection between a bypass graft vessel and the target coronary artery. As of March 31, 2017, the company had sold more than 15,200 C-Port Systems in the United States and Europe.
PAS-Port Proximal Anastomosis Systems (PAS-Port Systems) are used for proximal anastomosis, which is the connection between a bypass graft vessel and the aorta or other source of blood. As of March 31, 2017, the company had sold more than 46,600 PAS-Port systems in the United States, Europe, and Japan.
For the nine months ended March 31, 2017, the company generated revenue of $1.2 million related to the sale of automated anastomotic systems.
Market Overview
There is a growing demand for minimally invasive surgeries, which typically involve less pain, shorter hospital stays, and quicker recovery. Furthermore, the early detection of cancer is causing surgeons and patients to seek less invasive procedures to treat smaller lesions. According to estimates provided by the company, the global market for minimally invasive surgical stapling products totals $2 billion, and is expected to grow at an annual rate of eight percent through 2024.
Of the estimated 2.5 million surgical procedures performed annually in the United States (representing approximately 40 percent of the global market), the MicroCutter could be used in 280,000 procedures. This equates to an estimated market opportunity of $380 million in the United States alone.
Recent Developments
- On June 1, Dextera announced that it had filed a 510(k) with the FDA for expanded indications of use in liver, pancreas, kidney, and spleen surgery. These indications account for more than 80,000 procedures in the United States, and the company believes that the MicroCutter’s small size and increased articulation may contribute to less invasive surgeries. Surgeons in Europe are already successfully using the device for these applications.
- On May 16, Dextera closed an $8 million convertible preferred stock offering. The 8,000 Series B convertible preferred shares are each convertible into 3,704 shares of common stock at a conversion price of $0.27 per share. The preferred shares also include warrants to purchase additional shares at an exercise price of $0.27.
- In early May, Wedbush analyst Tao Levy reported that the Dextera was in active discussions with B. Braun / Aesculap regarding a strategic partnership agreement that would include global distribution of Dextera’s legacy cardiac products and expansion of the current distribution agreement for the MicroCutter in exchange for a private investment and a product development contract.
Third Quarter Earnings Review
Revenue for the quarter ended March 31, 2017, totaled $1.1 million compared with $1.9 million in the same period one year ago. The decline was primarily attributable to $1.4 million of license revenue from Intuitive Surgical recorded in the third quarter of 2016. This was partially offset by a $0.5 million increase in product sales. MicroCutter sales totaled $0.5 million, up from $0.3 million in the immediately preceding quarter.
Operating expenses increased 14 percent from the same period one year ago to $5.5 million due to greater product costs and research and development expenses. Dextera’s net loss for the third quarter of 2017 totaled $4.5 million, or $0.50 per share.
At March 31, 2017, the company reported cash and equivalents of $2.5 million, down from $12.7 million at June 30, 2016. Dextera also listed long-term debt of $3.4 million. In its most recent quarterly report, the company estimated that its current balance was sufficient to fund operations through May 2017. As noted above, the company subsequently secured financing with gross proceeds totaling $8 million.
Stock Influences
- Change in sales and adoption rates for the MicroCutter;
- Further regulatory approvals for the use of the MicroCutter;
- Licensing and royalty agreements for MicroCutter technology and reloadable cartridges; and
- M&A Activity.
Risk Factors
- The company is reliant on regulatory clearance and approval;
- The company is also reliant on sales of C-Port and PAS-Port systems to generate revenue;
- The company is currently producing the MicroCutter in small lots, and it may be unable to sufficiently scale up production;
- The company’s stock price is volatile and subject to speculation;
- The company is still burning cash, and may require additional financing in the future.
Stock Performance
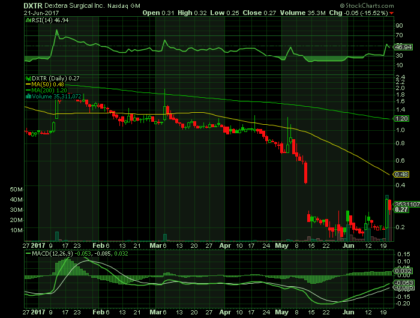
As of June 21, 2017, Dextera closed at $0.27, yielding a market capitalization of approximately $7 million. In the past 12 months, the stock has traded as high as $2.94 (in August 2016) but has recently traded as low as $0.17. The stock received a boost in January after the company issued strong corporate guidance, but is down more than 70 percent year-to-date.
In the past year, the annualized daily volatility was 137 percent. Daily trading volume has increased significantly in the past month, averaging 6.8 million shares per day.
Following are selected analyst ratings and price targets:
| Analyst | Firm | Rating | Price Target | Date |
| Charles Haff | Craig-Hallum | Hold | $0.70 | 5/19/2017 |
| Tao Levy | Wedbush | Outperform | $3.50 | 5/5/2017 |
Summary
Dextera has a strong product offering in the MicroCutter and has made good progress growing sales and adoption rates. Management has temporarily decreased its projections due to a supply chain disruption, but is optimistic that sales will continue to grow in the long-term and eventually overtake its legacy cardiac business. Furthermore, the company’s co-development project with Intuitive Surgical could produce an attractive revenue stream.
Still, Dextera is running low on cash, and will need to secure additional financing and/or strategic partnerships (such as licensing agreements) within the next year to keep the company afloat. The company will also need to increase its manufacturing capabilities to reach its desired scale.
***Get our small cap profiles, special situation and watch alerts in real time. We are now offering our VIP – SMS/text alert service for free, simply text the word “Traders” to the phone number “25827” from your cell phone***
Disclaimer
Traders News Source is a wholly owned subsidiary of Traders News Source LLC, herein referred to as TNS LLC.
Traders News Source has not been compensated for this report by anyone and the opinions if any are that of the author Ivan Neilson, CFA. Author’s Disclosure: I have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours. I, wrote this article myself, and it expresses my own opinions. I have no business relationship with any company whose stock is mentioned in the article.
This web site, published by TNS LLC, and is an investment newsletter that is built on the premise of assisting individual investors in learning about investing. Our goal as publishers of financial information is to provide research and analysis of investments to our subscribers. TNS LLC does not give buy or sell recommendations. We do purchase distribution rights from analyst, financial writers and bloggers for a fee that may be licensed to issue price targets and recommendations. Furthermore, we encourage you to speak to a licensed professional prior to making an investment in any type of publicly traded security.
We do sell advertising to other companies including brokerage firms, web sites, publicly traded issuers, investor relations firms, and investment publications, among others. TNS LLC makes no warranty as to the policies of these organizations, and in no way endorses their offers, services, or the content of their advertisements.
When an advertiser is a publicly traded company or a third party acting on behalf of a public company, we fully disclose all compensation in the email advertisement. Such disclosure is included in a disclosure statement in each of the advertisements sent via email.
17B Disclosure
Our reports/releases are a commercial advertisement and are for general information purposes ONLY. We are engaged in the business of marketing and advertising companies for monetary compensation. Never invest in any stock featured on our site or emails unless you can afford to lose your entire investment. The disclaimer is to be read and fully understood before using our services, joining our site or our email/blog list as well as any social networking platforms we may use.
PLEASE NOTE WELL: TNS LLC and its employees are not a Registered Investment Advisor, Broker Dealer or a member of any association for other research providers in any jurisdiction whatsoever.
Release of Liability: Through use of this website viewing or using you agree to hold TNS LLC, its operator’s owners and employees harmless and to completely release them from any and all liability due to any and all loss (monetary or otherwise), damage (monetary or otherwise), or injury (monetary or otherwise) that you may incur. The information contained herein is based on sources which we believe to be reliable but is not guaranteed by us as being accurate and does not purport to be a complete statement or summary of the available data. TNS LLC encourages readers and investors to supplement the information in these reports with independent research and other professional advice. All information on featured companies is provided by the companies profiled, or is available from public sources and TNS LLC makes no representations, warranties or guarantees as to the accuracy or completeness of the disclosure by the profiled companies. None of the materials or advertisements herein constitute offers or solicitations to purchase or sell securities of the companies profiled herein and any decision to invest in any such company or other financial decisions should not be made based upon the information provide herein. Instead TNS LLC strongly urges you conduct a complete and independent investigation of the respective companies and consideration of all pertinent risks. Readers are advised to review SEC periodic reports: Forms 10-Q, 10K, Form 8-K, insider reports, Forms 3, 4, 5 Schedule 13D.
TNS LLC is compliant with the Can Spam Act of 2003. TNS LLC does not offer such advice or analysis, and TNS LLC further urges you to consult your own independent tax, business, financial and investment advisors. Investing in micro-cap and growth securities is highly speculative and carries an extremely high degree of risk. It is possible that an investor’s investment may be lost or impaired due to the speculative nature of the companies profiled.
The Private Securities Litigation Reform Act of 1995 provides investors a ‘safe harbor’ in regard to forward-looking statements. Any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, goals, assumptions or future events or performance are not statements of historical fact may be “forward looking statements”. Forward looking statements are based on expectations, estimates, and projections at the time the statements are made that involve a number of risks and uncertainties which could cause actual results or events to differ materially from those presently anticipated. Forward looking statements in this action may be identified through use of words such as “projects”, “foresee”, “expects”, “will”, “anticipates”, “estimates”, “believes”, “understands”, or that by statements indicating certain actions & quote; “may”, “could”, or “might” occur.
Understand there is no guarantee past performance will be indicative of future results. In preparing this publication, TNS LLC has relied upon information supplied by its customers, publicly available information and press releases which it believes to be reliable; however, such reliability cannot be guaranteed. Investors should not rely on the information contained in this website. Rather, investors should use the information contained in this website as a starting point for doing additional independent research on the featured companies. The advertisements in this website are believed to be reliable, however, TNS LLC and its owners, affiliates, subsidiaries, officers, directors, representatives and agents disclaim any liability as to the completeness or accuracy of the information contained in any advertisement and for any omissions of materials facts from such advertisement. TNS LLC is not responsible for any claims made by the companies advertised herein, nor is TNS LLC responsible for any other promotional firm, its program or its structure.

