Xenetic Biosciences, Inc. (NASDAQ: XBIO), is a clinical-stage biopharmaceutical company, focused on the discovery, research, and development of next-generation biologic drugs and novel orphan oncology therapeutics. The company’s lead investigational product candidate is oncology therapeutic XBIO-101 (sodium cridanimod) for the treatment of progesterone resistant endometrial cancer.
On Monday, August 13, 2018, XBIO shares surged to an intra-day high of $4.60, a 44% jump from the previous Friday’s close. Share volume exceeded the daily average by a factor of ten.
The Company has entered into various research, development, license and supply agreements with Shire plc (“Shire”), Serum Institute of India (“Serum Institute”), its controlling stockholder PJSC Pharmsynthez (“Pharmsynthez”) and SynBio LLC (“SynBio”), which is now a wholly owned subsidiary of Pharmsynthez.
In 2018, an estimated 63,000 cases of endometrial cancer will be diagnosed in the U.S., and an estimated 11,000 disease-associated deaths will occur. While the incidence and mortality of endometrial cancer (EC) have been steadily increasing in the United States, the number of FDA-approved treatment options for late-stage EC remains extremely limited. In particular, there is a significant unmet need for therapies to treat advanced, recurrent, or metastatic EC.
Products
XBIO-101 The primary objective of the current open-label, multi-center, single-arm, two-period Phase 2 study is to assess the antitumor activity of XBIO-101 in conjunction with progestin therapy as measured by Overall Disease Control Rate in women with recurrent or persistent endometrial carcinoma not amenable to surgical treatment or radiotherapy who have either failed progestin monotherapy or who have been identified as PrR-negative. Interim Phase 2 data is expected in 2018.
The Company’s proprietary drug development platform, PolyXen, enables next-generation biologic drugs by improving their half-life and other pharmacological properties. The Company has ongoing business development activities to explore partnerships utilizing its PolyXen delivery platform.
XBIO-101 Phase II clinical trial
The primary objective of the open-label, multi-center, single-arm, two-period Phase 2 study is to assess the antitumor activity of XBIO-101 in conjunction with progestin therapy as measured by Overall Disease Control Rate in women with recurrent or persistent endometrial carcinoma not amenable to surgical treatment or radiotherapy who have either failed progestin monotherapy or who have been identified as PrR-negative.
Secondary objectives of the study are to assess progression-free survival, time to response, time to progression, duration of overall survival and Overall Disease Control Rate for subjects receiving XBIO-101 and progestin therapy; and to evaluate the safety and tolerability of XBIO-101 in conjunction with progestin therapy, as measured by adverse events, laboratory safety parameters, and cardiac safety assessments.
The study is expected to enroll a total of 72 women with recurrent or persistent endometrial cancer not amenable to surgical treatment or radiotherapy but suitable to be treated with progestin. All subjects determined to be PrR-negative at screening, as well as those subjects who experience disease progression after at least four weeks of progestin monotherapy, will receive XBIO-101 in combination with continued progestin treatment. Subjects will receive treatment until disease progression as defined according to RECIST 1.1 criteria.
About
Xenetic Biosciences, Inc., a clinical-stage biopharmaceutical company, focuses on the discovery, research, and development of next-generation biologic drugs and novel orphan oncology therapeutics. The company’s lead investigational product candidate is oncology therapeutic XBIO-101 (sodium cridanimod) for the treatment of progesterone resistant endometrial cancer. Its proprietary drug development platform, PolyXen, enables next-generation biologic drugs by improving their half-life and other pharmacological properties. The company has a research, development, license, and supply agreements with Shire plc, Serum Institute of India, PJSC Pharmsynthez, and SynBio LLC. Xenetic Biosciences, Inc. is headquartered in Lexington, Massachusetts.
Financial review Q2 2018
XBIO is a development stage biopharma company and has not generated revenues.
The Company incurred a net loss of approximately $3.8 million for the six months ended June 30, 2018. The Company had an accumulated deficit of approximately $149.8 million as of June 30, 2018 as compared to an accumulated deficit of approximately $145.9 million at December 31, 2017. Working capital was approximately $2.4 million as of June 30, 2018, and approximately $3.9 million as of December 31, 2017. The Company expects to continue incurring losses for the foreseeable future and will need to raise additional capital or pursue other strategic alternatives in the very near term in order to continue pursuit of its business plan and continue as a going concern.
Stock influences and risk factors
Any report of developments in the XBIO-101 Phase II trial could impact the company shares.
XBIO may require additional funding to achieve goals. Failure to obtain this capital when needed on acceptable terms, or at all, may force them to delay, limit or terminate product development efforts.
We may find it difficult to enroll patients in our clinical studies, which could delay or prevent clinical studies of our pharmaceutical products.
The business is substantially dependent on the success of clinical trials for XBIO-101 and the ability to achieve regulatory approval for the marketing of this product.
Stock chart
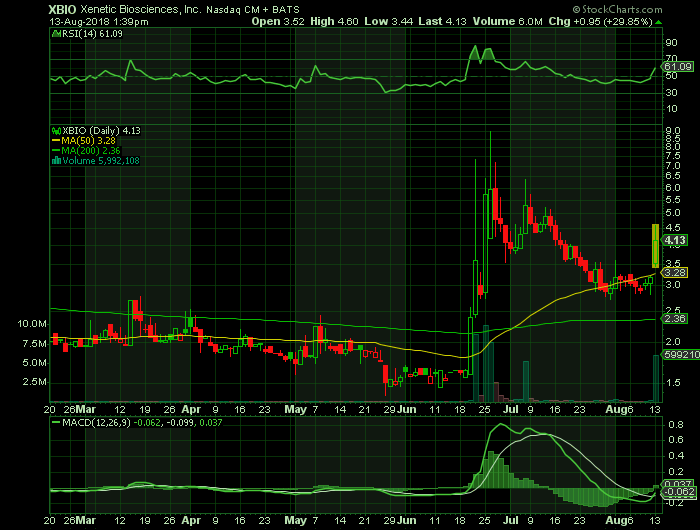
On Monday, August 13, 2018, XBIO was trading at $4.13 (+28.85%) on volume of 6.0 million shares. The current RSI (14) is 61.09
At $4.31, XBIO shares are trading above their 50 DMA and 200 DMA of $3.28 and $2.36 respectively.
Disclaimer
Traders News Source is a wholly owned subsidiary of Traders News Source LLC, herein referred to as TNS LLC.
Traders News Source has not been compensated for this report by anyone and the opinions if any are that of the author Vikas Agrawal, CFA. Author’s Disclosure: I have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours. I, wrote this article myself, and it expresses my own opinions. I have no business relationship with any company whose stock is mentioned in the article.
This web site, published by TNS LLC, and is an investment newsletter that is built on the premise of assisting individual investors in learning about investing. Our goal as publishers of financial information is to provide research and analysis of investments to our subscribers. TNS LLC does not give buy or sell recommendations. We do purchase distribution rights from analyst, financial writers and bloggers for a fee that may be licensed to issue price targets and recommendations. Furthermore, we encourage you to speak to a licensed professional prior to making an investment in any type of publicly traded security.
We do sell advertising to other companies including brokerage firms, web sites, publicly traded issuers, investor relations firms, and investment publications, among others. TNS LLC makes no warranty as to the policies of these organizations, and in no way endorses their offers, services, or the content of their advertisements.
When an advertiser is a publicly traded company or a third party acting on behalf of a public company, we fully disclose all compensation in the email advertisement. Such disclosure is included in a disclosure statement in each of the advertisements sent via email.
17B Disclosure
Our reports/releases are a commercial advertisement and are for general information purposes ONLY. We are engaged in the business of marketing and advertising companies for monetary compensation. Never invest in any stock featured on our site or emails unless you can afford to lose your entire investment. The disclaimer is to be read and fully understood before using our services, joining our site or our email/blog list as well as any social networking platforms we may use.
PLEASE NOTE WELL: TNS LLC and its employees are not a Registered Investment Advisor, Broker Dealer or a member of any association for other research providers in any jurisdiction whatsoever.
Release of Liability: Through use of this website viewing or using you agree to hold TNS LLC, its operator’s owners and employees harmless and to completely release them from any and all liability due to any and all loss (monetary or otherwise), damage (monetary or otherwise), or injury (monetary or otherwise) that you may incur. The information contained herein is based on sources which we believe to be reliable but is not guaranteed by us as being accurate and does not purport to be a complete statement or summary of the available data. TNS LLC encourages readers and investors to supplement the information in these reports with independent research and other professional advice. All information on featured companies is provided by the companies profiled, or is available from public sources and TNS LLC makes no representations, warranties or guarantees as to the accuracy or completeness of the disclosure by the profiled companies. None of the materials or advertisements herein constitute offers or solicitations to purchase or sell securities of the companies profiled herein and any decision to invest in any such company or other financial decisions should not be made based upon the information provide herein. Instead TNS LLC strongly urges you conduct a complete and independent investigation of the respective companies and consideration of all pertinent risks. Readers are advised to review SEC periodic reports: Forms 10-Q, 10K, Form 8-K, insider reports, Forms 3, 4, 5 Schedule 13D.
TNS LLC is compliant with the Can Spam Act of 2003. TNS LLC does not offer such advice or analysis, and TNS LLC further urges you to consult your own independent tax, business, financial and investment advisors. Investing in micro-cap and growth securities is highly speculative and carries an extremely high degree of risk. It is possible that an investor’s investment may be lost or impaired due to the speculative nature of the companies profiled.
The Private Securities Litigation Reform Act of 1995 provides investors a ‘safe harbor’ in regard to forward-looking statements. Any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, goals, assumptions or future events or performance are not statements of historical fact may be “forward looking statements”. Forward looking statements are based on expectations, estimates, and projections at the time the statements are made that involve a number of risks and uncertainties which could cause actual results or events to differ materially from those presently anticipated. Forward looking statements in this action may be identified through use of words such as “projects”, “foresee”, “expects”, “will”, “anticipates”, “estimates”, “believes”, “understands”, or that by statements indicating certain actions & quote; “may”, “could”, or “might” occur.
Understand there is no guarantee past performance will be indicative of future results. In preparing this publication, TNS LLC has relied upon information supplied by its customers, publicly available information and press releases which it believes to be reliable; however, such reliability cannot be guaranteed. Investors should not rely on the information contained in this website. Rather, investors should use the information contained in this website as a starting point for doing additional independent research on the featured companies. The advertisements in this website are believed to be reliable, however, TNS LLC and its owners, affiliates, subsidiaries, officers, directors, representatives and agents disclaim any liability as to the completeness or accuracy of the information contained in any advertisement and for any omissions of materials facts from such advertisement. TNS LLC is not responsible for any claims made by the companies advertised herein, nor is TNS LLC responsible for any other promotional firm, its program or its structure.


