Co-Diagnostics, Inc. (NASDAQ: CODX), is a molecular diagnostics company that intends to manufacture and sell reagents used for diagnostic tests that function via the detection and/or analysis of nucleic acid molecules. It also intends to sell diagnostic equipment from other manufacturers as self-contained lab systems.
It’s been almost a year since CODX completed its IPO raising $7 million with an offering of 1.2 million shares at $6 a share. In late February 2018 the company shares hit their 2018 low of $1.45 per share.
On Friday, June 1, 2018, CODX shares closed at $4.79 (+65.17%) on unusually high volume of 20.9 million shares traded.
Recent Events
June 1, 2018. Co-Diagnostics announced that the Company’s CoPrimer™ technology is being employed to achieve the objectives of a research project conducted by the Wang Group at Stanford University. The purpose of the study is to develop more efficient methods to detect multiple genetic mutations in the EGFR (epidermal growth factor receptor) gene, including the most frequently occurring mutations in lung cancer, within a single reaction. https://finance.yahoo.com/news/co-diagnostics-inc-announces-participation-103000825.html
Products
Co-Diagnostics’ CoDx™ portfolio of molecular diagnostics development products and tests represent a radical new advancement in the understanding of the molecular interactions of DNA. Leading a global health revolution through mathematical innovations, cost reductions, and wider margins with our shareholders in mind,
- using highly specialized, proprietary cooperative-theory mathematics,
- leading to a revolutionary leap forward in the detection of infectious diseases, genetic disorders and other conditions,
- at a fraction of the cost of other DNA-based tests,
- designed for a new generation of affordable, mobile point-of-care diagnostic devices, and compatible with many other existing, commercially-available devices,
- making state-of-the-art diagnostics available anywhere in the world, including developing countries.
CoPrimers™ are sixth-generation probes for real-time PCR and are the latest innovation from Dr. Brent Satterfield and Co-Diagnostics, providing the most potent and advanced reagents available today. CoPrimers™ enhance the speed, accuracy and cost effectiveness of PCR. CoPrimers™ virtually eliminate the formation of primer dimers, a bane to test designers, which occur when errors are amplified during the PCR process, causing false positives and negatives. CoPrimers™ are most effective at eliminating primer dimers when running multiple PCR tests simultaneously (multiplexing).
RapidProbe™ is a fifth- generation mathematically-engineered probe design for real-time PCR. It increases probe accuracy while reducing design complexity and expense. RapidProbe™ is faster than fourth generation Tentacle Probes™ and far less expensive and up to 200 times faster than hairpin probes. Highly sensitive, RapidProbe™ can detect even a single virus in a DNA sample. Brighter probes translate to higher accuracy and lower-cost detection equipment. RapidProbe™ brings the quencher and flourophore into close proximity. When the probe initiates hybridization, it “unzips,” creating a brighter signal than standard probes.
HotStaRT™ – Reduces false readings in real-time PCR. HotStaRT™ is a unique hot start for PCR and is the first to work on both RNA and DNA reactions, reducing both false positives and negatives. HotStaRT™ adds proprietary chemical “hooks” to an oligonucleotide, causing them to stick to the binding site at low temperatures, reducing non-specific reactions. HotStaRT™ works on both polymerase and reverse transcriptase reactions. The charts below illustrate its effectiveness at improving sensitivity and outcomes on the reverse transcriptase reaction.
About
Co-Diagnostics, Inc., a molecular diagnostics company, intends to manufacture and sell reagents used for diagnostic tests that function via the detection and/or analysis of nucleic acid molecules. It also intends to sell diagnostic equipment from other manufacturers as self-contained lab systems. Co-Diagnostics, Inc. was founded in 2013 and is headquartered in Salt Lake City, Utah.
Working in tandem, both affordable tests, and affordable diagnostic devices will revolutionize DNA diagnostics, for the first time making accurate detection of genetic disorders and infectious diseases far less expensive and much more broadly available. This is made possible by combining proprietary software, patented reagent technologies and an array of affordable, accurate PCR tests.
Dr. Brent Satterfield invented the Company’s suite of intellectual properties. CODX scientists were the first to understand the complex mathematics of DNA test design, to “engineer” a DNA test and to automate algorithms that rapidly screen millions of possible options to pinpoint the optimum design. This bio-engineering approach to DNA test development is unique to Co-Diagnostics and is based on “cooperative theory,” a new generation of mathematical analytics created by Dr. Satterfield.
CDI’s diagnostics systems enable very rapid, low-cost, sophisticated molecular testing for organisms and genetic diseases by greatly automating historically complex procedures in both the development and administration of tests. CDI’s newest technical advance involves a novel approach to PCR primer design (cooperative primers) that eliminates one of the key vexing issues of PCR amplification, the exponential growth of primer-dimer pairs which adversely interferes with identification of the target DNA. In addition, CDI scientists have enhanced the understanding of the mathematics of DNA test design, so as to “engineer” a DNA test and automate algorithms to screen millions of possible designs to find the optimum DNA test design. CDI’s proprietary platform of Co-Dx™ technologies integrates and streamlines these steps as it analyzes biological samples.
Co-Diagnostics’ CoDx™ portfolio of molecular diagnostics development products and tests represent a radical new advancement in the understanding of the molecular interactions of DNA. The Company uses highly specialized, proprietary cooperative-theory mathematics, leading to a revolutionary leap forward in the detection of infectious diseases, genetic disorders and other conditions. CoDx™ tests are a fraction of the cost of other DNA-based tests, designed for a new generation of affordable, mobile point-of-care diagnostic devices and compatible with many other devices, making state-of-the-art diagnostics available anywhere in the world, including developing countries.
Financial review FYE 2017 and Q1 2018
Revenue for the year totaled $7,700, primarily licensing revenue, compared to zero revenue in full year 2016.
Deferred income at December 31, 2017, amounted to $184,000; this is revenue that will be recognized in future quarters.
The Company ended the year with cash and equivalents of $3.5 million and zero long-term debt.
The Company reported a net loss for full year 2017 of approximately $7.0 million, or $0.63 per fully diluted share. This included a non-cash loss of $2,072,000 that was recognized when noteholders converted debt to stock in the IPO, and $2,033,000 of non-cash expenses related to the issuance of stock for services.
Q1 2018 financial review
For the three months ended March 31, 2018 we generated $9,696 of revenues compared to no revenues in the three months ended March 31, 2017.
For the three months ended March 31, 2018 and for the three months ended March 31, 2017, they recorded no costs of revenues.
Operating expenses were $1,287,127 for the three months ended March 31, 2018 compared to total operating expenses of $564,365 for the three months ended March 31, 2017.
Research and development expenses increased by $31,727 from $265,688 for the three months ended March 31, 2017 to $297,415 for the three months ended March 31, 2018.
Net loss for the three months ended March 31, 2018 of $1,310,233 compared with a net loss for the three months ended March 31, 2017 of $705,742.
At March 31, 2018, they had cash and cash equivalents of $2,541,242.
Stock influences and risk factors
If the potential market for the CODX products grows substantially, it could act as a catalyst for the company’s shares;
The company may require additional funding to operate and develop products and that financing could be dilutive;
The biotech industry carries inherent risks such as regulatory restrictions and intense competition;
The company has produced limited revenues and only net losses and there is no guarantee they ever will be profitable.
Stock chart
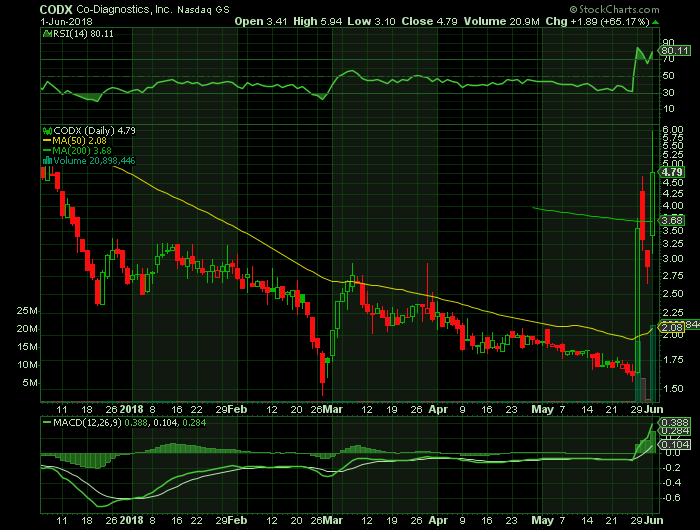
On Friday, June 1, 2018, CODX shares closed at $4.79 (+65.17%) on unusually high volume of 20.9 million shares traded. The current RSI (14) is 80.11
At $4.79, CODX shares are trading above their 50 DMA and 200 DMA of $2.08 and $3.68 respectively.

