Unum Therapeutics Inc. (NASDAQ: UMRX), a clinical-stage biopharmaceutical company focused on the development of cellular immunotherapies based on its novel, universal Antibody-Coupled T-cell Receptor (ACTR) technology platform.
On May 14th, the company announced financial results for the quarter ended March 31, 2018, following its successful initial public offering in April 2018 and concurrent private placement, the company is in a strong financial position to continue developing its proprietary, universal ACTR technology platform and rapidly advancing pipeline of cellular immunotherapies through clinical development.
Key highlights of the first quarter:
- Successfully Completed IPO and Concurrent Private Placement Raising $77 Million in Gross Proceeds
- Initiated Cohort Expansion of ATTCK-20-2 Phase I Trial of ACTR087 in Combination with Rituximab in Patients with CD20+ r/r Non-Hodgkin Lymphoma; Updated Data Expected in 4Q 2018
- Phase I Trials of ACTR087 in Combination with SEA-BCMA in Patients with r/r Multiple Myeloma and ACTR707 in Combination with Rituximab in Patients with CD20+ r/r Non-Hodgkin Lymphoma Ongoing; Preliminary Data from Both Trials Expected in 4Q 2018
- On Track to File IND for First Solid Tumor Program, ACTR707 in Combination with Trastuzumab in Patients with HER2+ Advanced Malignancies, in 2H 2018
UMRX is currently evaluating the potential of ACTR in combination with different tumor-targeting antibodies, in three ongoing multi-centres Phase I trials, ATTCK-20-2 and ATTCK-20-03 evaluating ACTR087 and ACTR707, respectively, in combination with rituximab in patients with CD20+ r/r Non-Hodgkin Lymphoma (NHL), and ATTCK-17-01 evaluating ACTR087 in combination with SEA-BCMA in patients with r/r multiple myeloma.
Upcoming Milestone:
The company expects to report preliminary data from these three trials late this year. In the second half of 2018, it is also looking forward to filing an IND and initiating clinical development of ACTR707 in combination with trastuzumab for the treatment of patients with HER2+ advanced cancers, UMRX’ first solid tumor product candidate.
Analyst tracking the stock believes that UMRX continues to make sustained progress in its field, most of which could lead them to success. Also, Company’s healthy financial position, as well as promising business profile, makes it a compelling investment/trading option.
The company is presently featuring as “Strong Buy” and the average target price is around $20 with a high estimate of approximately $21.00 and a low range of $18.00. On the flip side, the company is exposed to the risk associated with any biotech play.
About the Company: Unum Therapeutics is a clinical-stage biopharmaceutical company focused on the development and commercialization of novel immunotherapy products designed to harness the power of a patient’s immune system to cure cancer. Unum’s novel proprietary technology, antibody-coupled T cell receptor (ACTR), is a universal, engineered cell therapy intended to be used in combination with a wide range of tumor-specific antibodies to target different tumor types. Unum is actively building a pipeline of product candidates composed of ACTR T cells co-administered with antibodies for use in both hematologic and solid tumor cancers. The Company is headquartered in Cambridge, MA.
Recent developments in details:
- Initiated Cohort Expansion Phase of ATTCK-20-2 Phase I trial; Plans to Expand Clinical Development: In May 2018, Unum initiated the cohort expansion phase of the ATTCK-20-2 trial evaluating safety and anti-lymphoma activity of ACTR087 at the preliminary recommended phase 2 dose (RP2D) level used in combination with rituximab in patients with CD20+ r/r NHL. Unum expects to report updated data, including preliminary data from this phase of the ATTCK-20-2 trial, in the fourth quarter of 2018.
These data will also inform the strategy for a planned multi-center Phase II clinical trial exploring ACTR T cells used in combination with rituximab in patients with CD20+ r/r NHL who received prior CD19 CAR T cell therapy.
Besides, Unum intends to file a protocol amendment to the ATTCK-20-2 trial in the second half of 2018 to explore ACTR087 in combination with an alternative rituximab dosing regimen from that currently being studied.
- Initiated Patient Enrollment and Dosing in ATTCK-17-01 Phase I trial: In the first quarter, Unum initiated patient enrollment in ATTCK-17-01, a Phase I, multi-center, an open-label clinical trial designed to test the safety, tolerability, and anti-myeloma activity of ACTR087 used in combination with SEA-BCMA in patients with r/r multiple myeloma. Unum is currently enrolling and dosing patients in this trial and expects to report preliminary data in the fourth quarter of 2018.
- Continued Enrollment in ATTCK-20-03 Phase I trial: In the fourth quarter of 2017, Unum initiated patient enrollment in Phase I, multi-center, an open-label clinical trial called ATTCK-20-03, evaluating the safety, tolerability, and anti-lymphoma activity of ACTR707 used in combination with rituximab in patients with CD20+ r/r NHL. Unum has completed enrollment in the first dose level of this ongoing dose escalation study and expects to report preliminary data from the trial in the fourth quarter of 2018.
- On Track to File IND for First Solid Tumor ACTR Product Candidate in the Second Half of 2018: Unum is on track to file an IND in the second half of 2018 for ACTR707 in combination with trastuzumab for the treatment of patients with HER2+ advanced cancers.
First Quarter 2018 Results:
Revenue: Collaboration revenue recognized during the three months ended March 31, 2018 and 2017, of $2.2 million and $1.8 million, respectively, reflects the recognition of a portion of the $25.0 million upfront payment received from Seattle Genetics under Unum’s collaboration agreement as well as reimbursements of research and development costs by Seattle Genetics. Effective January 1, 2018, Unum adopted the new revenue recognition standard, ASC 606, which changed the manner in which the Company recognizes revenue from this collaboration agreement.
Net Loss: Net loss attributable to common stockholders was $6.8 million, or $0.66 per share, for the three months ended March 31, 2018, and $6.0 million, or $0.58 per share, for the three months ended March 31, 2017.
Cash, Cash Equivalents, and Marketable Securities: As of March 31, 2018, Unum had cash, cash equivalents, and marketable securities of $32.4 million. The Company believes that the net proceeds from the IPO and concurrent private placement, together with its existing cash, cash equivalents, and marketable securities, will fund operating expenses and capital expenditure requirements through at least December 2019, without considering available borrowings under its loan and security agreement.
Key risk factors and potential stock drivers:
- Successful completion of the upcoming milestones would lead future direction for the company. Any adversities related to these future milestones might adversely impact the overall investor sentiments.
- UMRX is still an early stage entity and has not yet generated revenue and will likely operate at a loss as it grows its market position and seeks ways to monetize it. The company’ prospects are significantly dependent on BPX-01 and BPX-04, which might have limited sales potential initially.
- UMRX has a history of operating losses. Therefore, any time or cost overrun in its ongoing R&D activities and its impact on business & financial profile will remain a key business sensitivity factor.
Stock Chart:
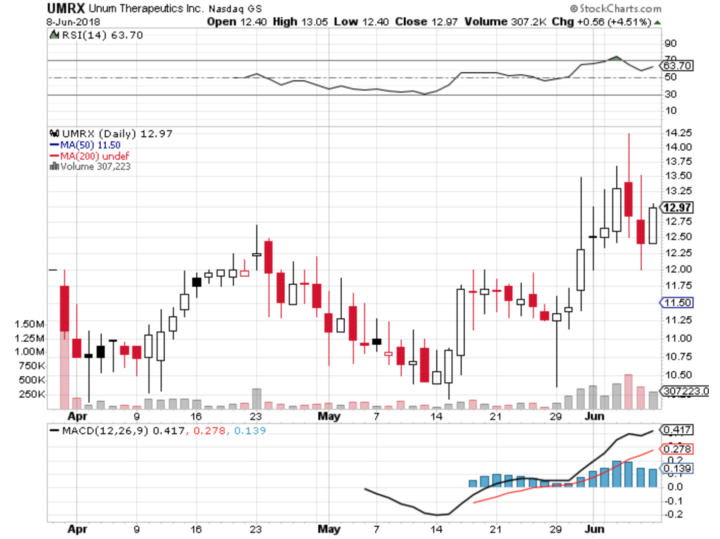
Comments:
- On Friday, June 8th, 2018, UMRX closed at $12.97, on an average volume of 167,016 million shares exchanging hands. Market capitalization is $387 million. The current RSI is at 63.70
- In the past 52 weeks, shares of UMRX have traded as low as $10.15 and as high as $14.24.
- At $12.97, shares of UMRX are trading above its 50-day moving average (MA) at $11.50
- The present support and resistance levels for the stock are at $12.16 & $13.45 respectively.
Disclaimer
Traders News Source is a wholly owned subsidiary of Traders News Source LLC, herein referred to as TNS LLC.
Traders News Source has not been compensated for this report by anyone and the opinions if any are that of the author Vikas Agrawal, CFA. Author’s Disclosure: I have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours. I, wrote this article myself, and it expresses my own opinions. I have no business relationship with any company whose stock is mentioned in the article.
This web site, published by TNS LLC, and is an investment newsletter that is built on the premise of assisting individual investors in learning about investing. Our goal as publishers of financial information is to provide research and analysis of investments to our subscribers. TNS LLC does not give buy or sell recommendations. We do purchase distribution rights from analyst, financial writers and bloggers for a fee that may be licensed to issue price targets and recommendations. Furthermore, we encourage you to speak to a licensed professional prior to making an investment in any type of publicly traded security.
We do sell advertising to other companies including brokerage firms, web sites, publicly traded issuers, investor relations firms, and investment publications, among others. TNS LLC makes no warranty as to the policies of these organizations, and in no way endorses their offers, services, or the content of their advertisements.
When an advertiser is a publicly traded company or a third party acting on behalf of a public company, we fully disclose all compensation in the email advertisement. Such disclosure is included in a disclosure statement in each of the advertisements sent via email.
17B Disclosure
Our reports/releases are a commercial advertisement and are for general information purposes ONLY. We are engaged in the business of marketing and advertising companies for monetary compensation. Never invest in any stock featured on our site or emails unless you can afford to lose your entire investment. The disclaimer is to be read and fully understood before using our services, joining our site or our email/blog list as well as any social networking platforms we may use.
PLEASE NOTE WELL: TNS LLC and its employees are not a Registered Investment Advisor, Broker Dealer or a member of any association for other research providers in any jurisdiction whatsoever.
Release of Liability: Through use of this website viewing or using you agree to hold TNS LLC, its operator’s owners and employees harmless and to completely release them from any and all liability due to any and all loss (monetary or otherwise), damage (monetary or otherwise), or injury (monetary or otherwise) that you may incur. The information contained herein is based on sources which we believe to be reliable but is not guaranteed by us as being accurate and does not purport to be a complete statement or summary of the available data. TNS LLC encourages readers and investors to supplement the information in these reports with independent research and other professional advice. All information on featured companies is provided by the companies profiled or is available from public sources and TNS LLC makes no representations, warranties or guarantees as to the accuracy or completeness of the disclosure by the profiled companies. None of the materials or advertisements herein constitute offers or solicitations to purchase or sell securities of the companies profiled herein and any decision to invest in any such company or other financial decisions should not be made based upon the information provide herein. Instead TNS LLC strongly urges you conduct a complete and independent investigation of the respective companies and consideration of all pertinent risks. Readers are advised to review SEC periodic reports: Forms 10-Q, 10K, Form 8-K, insider reports, Forms 3, 4, 5 Schedule 13D.
TNS LLC is compliant with the Can Spam Act of 2003. TNS LLC does not offer such advice or analysis, and TNS LLC further urges you to consult your own independent tax, business, financial and investment advisors. Investing in micro-cap and growth securities is highly speculative and carries an extremely high degree of risk. It is possible that an investor’s investment may be lost or impaired due to the speculative nature of the companies profiled.
The Private Securities Litigation Reform Act of 1995 provides investors a ‘safe harbor’ in regard to forward-looking statements. Any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, goals, assumptions or future events or performance are not statements of historical fact may be “forward looking statements”. Forward looking statements are based on expectations, estimates, and projections at the time the statements are made that involve a number of risks and uncertainties which could cause actual results or events to differ materially from those presently anticipated. Forward looking statements in this action may be identified through use of words such as “projects”, “foresee”, “expects”, “will”, “anticipates”, “estimates”, “believes”, “understands”, or that by statements indicating certain actions & quote; “may”, “could”, or “might” occur.
Understand there is no guarantee past performance will be indicative of future results. In preparing this publication, TNS LLC has relied upon information supplied by its customers, publicly available information and press releases which it believes to be reliable; however, such reliability cannot be guaranteed. Investors should not rely on the information contained in this website. Rather, investors should use the information contained in this website as a starting point for doing additional independent research on the featured companies. The advertisements in this website are believed to be reliable, however, TNS LLC and its owners, affiliates, subsidiaries, officers, directors, representatives and agents disclaim any liability as to the completeness or accuracy of the information contained in any advertisement and for any omissions of materials facts from such advertisement. TNS LLC is not responsible for any claims made by the companies advertised herein, nor is TNS LLC responsible for any other promotional firm, its program or its structure.

