Company Overview
AbbVie Inc. (NYSE: ABBV) is a research-based pharmaceutical company. AbbVie is engaged in the discovery, development, manufacturing, and sale of a variety of pharmaceutical products. These include treatments for chronic autoimmune diseases, oncology, virology, neurological disorders, and metabolic diseases. The company also maintains an active development pipeline in these areas, with additional targeted investment in cystic fibrosis and women’s health.
AbbVie’s products are generally sold to wholesalers, distributors, government agencies, health care facilities, and retailers. The company was originally an integrated pharmaceutical unit of Abbott Laboratories, but was spun off in January 2013. AbbVie is headquartered in North Chicago, Illinois.
Products
AbbVie’s primary source of revenue is Humira (adalimumab), but the company has several other products which have been approved for sale. Below is a summary of the company’s product portfolio and its annual net revenues:

Source: Company Reports
As shown above, AbbVie’s three leading products contributed more than 75 percent of the company’s net revenues in 2016.
Humira is one of the best-selling drugs in the world by a significant margin. It is a biologic therapy administered as a subcutaneous injection for the treatment of (among other things) rheumatoid arthritis, psoriatic arthritis, Crohn’s disease, plaque psoriasis, ulcerative colitis, and other autoimmune diseases. While AbbVie faces significant potential competition from manufacturers of biosimilar products, the company believes that it can enforce its intellectual property rights until 2022.
Imbruvica (ibrutinib) is a once-daily oral therapy for the treatment of patients with chronic lymphocytic leukemia. It has also received conditional approval for the treatment of mantle cell lymphoma and marginal cell lymphoma. Imbruvica has also received breakthrough therapy designation for chronic graft-versus-host-disease. While Imbruiva brings in significantly less revenue than Humira, it is growing rapidly. It is currently in three Phase II studies and three Phase III studies for a variety of additional applications. With further regulatory approvals, the company believes that annual net revenue from Imbruvica could hit $7 billion.
Viekira (ombitasvir, paritaprevir and ritonavir tablets, dasabuvir tablets) is an all-oral, short-course, therapy for the treatment of genotype 1 chronic hepatitis C virus (HCV). In Europe, the company’s HCV treatment is marketed as Viekira + Exviera and is approved for use in patients with genotype 1 and genotype 4 HCV. Sales of Viekira fell in 2016, likely due to several new HCV treatments becoming available.
Pipeline
AbbVie’s pipeline consists of more than 50 compounds or indications in clinical development individually or under collaboration / licensing agreements. More than half of these programs are in mid or late-stage development. At a recent healthcare conference hosted by Deutsche Bank, AbbVie CFO Bill Chase stated that the company expects to have 20 new assets in place by 2020. Some of the more promising candidates are:
Rova-T – an oncology treatment in registrational trials for small cell lung cancer and in early-stage clinical development for other solid tumors. If successful, the drug could hit peak annual sales of $5 billion.
Risankizumab – an anti-IL-23 monoclonal biologic antibody in Phase III development for psoriasis. It is also being evaluated for therapy in Crohn’s disease, psoriatic arthritis, and asthma. If successful, the drug could hit peak annual sales of $4 billion.
Upadacitinib (ABT-494) – is the company’s selective JAK-1 inhibitor which is currently in late-stage development for the treatment of rheumatoid arthritis. Projected peak sales for upadacitinib total $3.5 billion annually.
Upcoming Study Results
AbbVie is scheduled to report on several clinical studies in the next two quarters, and the results could provide an immediate boost to its share price. For instance, the TRINITY pivotal study of Rova-T for the treatment of small cell lung cancer is expected sometime in the second half of 2017, as is Phase III data for risankizumab as a treatment for psoriasis. With respect to upadacitinib, the SELECT-BEYOND study involving rheumatoid arthritis patients is expected to read out sometime this summer, and another Phase III study (SELECT-COMPARE) using Humira as a comparison drug should be reported on by the end of the year.
First Quarter Earnings Review
AbbVie reported 2017 first quarter revenue of $6.5 billion, representing 10 percent growth year-over-year. This was driven by strong sales of Humira and Imbruvica which helped to overcome falling sales from Viekira and certain other drugs. Global Humira sales increased 15 percent year-over-year, while net revenue from Imbruvica increased 45 percent to $551 million. Overall, the company reported a gross margin of 75 percent on a GAAP basis, and nearly 80 percent on an adjusted basis.
Selling, general, and administrative expenses were roughly flat, while research and development expenses increased 20 percent to $1.1 billion. Overall, the company’s operating margin decreased slightly to 37 percent. Adjusted diluted earnings per share (which excludes intangible asset amortization and certain other items) was $1.28 per share, an 11 percent increase from the same period one year ago.
For the quarter ended March 31, 2017, the company had cash flows from operations of $2.1 billion, comparable to the same period last year. Approximately $1 billion was paid out as dividends while another $900 million was used to purchase treasury stock.
At March 31, 2017, AbbVie listed cash and short-term investments equal to $6.1 billion, slightly lower than the previous quarter’s balance of $6.4 billion. The company had net working capital of $6.7 billion, and listed long-term debt and lease obligations of $36.5 billion.
Stock Influences
- Clinical trial results from the company’s pipeline products (Rova-T, risankizumab upadacitinib);
- Clinical trial results from the company’s existing product lines (Imbruvica);
- Successful challenges to Humira’s intellectual property; and
- Acquisition and/or licensing of new technology.
Risk Factors
- Humira accounted for more than 60 percent of the company’s revenue in 2016, and it is uncertain whether any of its existing candidates will be able to replace these sales if/when the company faces more intense competition from biosimilars;
- Furthermore, any events that adversely affect Humira would have a material impact on the company’s results;
- The company faces competition from biosimilars for many other products; and
- Due to the nature of its business, AbbVie is subject to significant government regulation.
Stock Performance
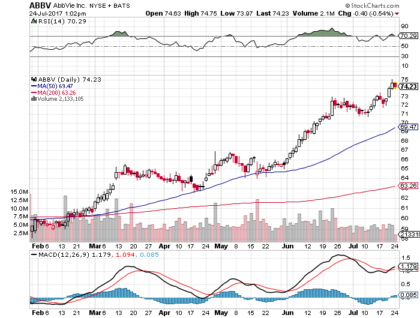
As of June 23, 2017, shares of AbbVie closed at $72.64, yielding a market capitalization of approximately $116 billion. In the past year, the shares have gained more than 20 percent. The stock price sold off sharply prior to the U.S election, hitting a low of $55.06, but have since recovered and recently hit a one-year high of $71.81.
The stock pays a quarterly dividend $0.64 per share which equates to an annual yield of approximately 3.6 percent. AbbVie has raised its dividend every year since inception, and in February the company announced a $5 billion increase to its stock repurchase program.
Following are selected analyst ratings and price targets:
| Analyst | Firm | Rating | Price Target | Date |
| John Boris | SunTrust | Buy | $85.00 | 6/11/2017 |
| Jeffrey Holford | Jeffries | Buy | $90.00 | 6/9/2017 |
| Matthew Harrison | Morgan Stanley | Equal-Weight | $68.00 | 6/8/2017 |
Summary
AbbVie has a dominant product in Humira that is likely to continue its growth trajectory. However, it accounts for more than 60 percent of the company’s revenue, and this lack of diversification is worrying to investors. If competitors successfully challenge Humira’s intellectual property and begin marketing biosimilars on a large scale, the company could be adversely affected.
However, AbbVie is confident that it can defend Humira until 2022, giving the company time to develop new products and find additional applications for existing drugs. With several new product candidates and many ongoing clinical trials, the company is well-positioned to diversify its product base. In the interim, AbbVie has a strong history of rewarding shareholders with dividends and buybacks and generates the necessary cash flows to continue doing so.
***Get our small cap profiles, special situation and watch alerts in real time. We are now offering our VIP – SMS/text alert service for free, simply text the word “Traders” to the phone number “25827” from your cell phone***
Disclaimer
Traders News Source is a wholly owned subsidiary of Traders News Source LLC, herein referred to as TNS LLC.
Traders News Source has not been compensated for this report by anyone and the opinions if any are that of the author Ivan Neilson, CFA. Author’s Disclosure: I have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours. I, wrote this article myself, and it expresses my own opinions. I have no business relationship with any company whose stock is mentioned in the article.
This web site, published by TNS LLC, and is an investment newsletter that is built on the premise of assisting individual investors in learning about investing. Our goal as publishers of financial information is to provide research and analysis of investments to our subscribers. TNS LLC does not give buy or sell recommendations. We do purchase distribution rights from analyst, financial writers and bloggers for a fee that may be licensed to issue price targets and recommendations. Furthermore, we encourage you to speak to a licensed professional prior to making an investment in any type of publicly traded security.
We do sell advertising to other companies including brokerage firms, web sites, publicly traded issuers, investor relations firms, and investment publications, among others. TNS LLC makes no warranty as to the policies of these organizations, and in no way endorses their offers, services, or the content of their advertisements.
When an advertiser is a publicly traded company or a third party acting on behalf of a public company, we fully disclose all compensation in the email advertisement. Such disclosure is included in a disclosure statement in each of the advertisements sent via email.
17B Disclosure
Our reports/releases are a commercial advertisement and are for general information purposes ONLY. We are engaged in the business of marketing and advertising companies for monetary compensation. Never invest in any stock featured on our site or emails unless you can afford to lose your entire investment. The disclaimer is to be read and fully understood before using our services, joining our site or our email/blog list as well as any social networking platforms we may use.
PLEASE NOTE WELL: TNS LLC and its employees are not a Registered Investment Advisor, Broker Dealer or a member of any association for other research providers in any jurisdiction whatsoever.
Release of Liability: Through use of this website viewing or using you agree to hold TNS LLC, its operator’s owners and employees harmless and to completely release them from any and all liability due to any and all loss (monetary or otherwise), damage (monetary or otherwise), or injury (monetary or otherwise) that you may incur. The information contained herein is based on sources which we believe to be reliable but is not guaranteed by us as being accurate and does not purport to be a complete statement or summary of the available data. TNS LLC encourages readers and investors to supplement the information in these reports with independent research and other professional advice. All information on featured companies is provided by the companies profiled, or is available from public sources and TNS LLC makes no representations, warranties or guarantees as to the accuracy or completeness of the disclosure by the profiled companies. None of the materials or advertisements herein constitute offers or solicitations to purchase or sell securities of the companies profiled herein and any decision to invest in any such company or other financial decisions should not be made based upon the information provide herein. Instead TNS LLC strongly urges you conduct a complete and independent investigation of the respective companies and consideration of all pertinent risks. Readers are advised to review SEC periodic reports: Forms 10-Q, 10K, Form 8-K, insider reports, Forms 3, 4, 5 Schedule 13D.
TNS LLC is compliant with the Can Spam Act of 2003. TNS LLC does not offer such advice or analysis, and TNS LLC further urges you to consult your own independent tax, business, financial and investment advisors. Investing in micro-cap and growth securities is highly speculative and carries an extremely high degree of risk. It is possible that an investor’s investment may be lost or impaired due to the speculative nature of the companies profiled.
The Private Securities Litigation Reform Act of 1995 provides investors a ‘safe harbor’ in regard to forward-looking statements. Any statements that express or involve discussions with respect to predictions, expectations, beliefs, plans, projections, objectives, goals, assumptions or future events or performance are not statements of historical fact may be “forward looking statements”. Forward looking statements are based on expectations, estimates, and projections at the time the statements are made that involve a number of risks and uncertainties which could cause actual results or events to differ materially from those presently anticipated. Forward looking statements in this action may be identified through use of words such as “projects”, “foresee”, “expects”, “will”, “anticipates”, “estimates”, “believes”, “understands”, or that by statements indicating certain actions & quote; “may”, “could”, or “might” occur.
Understand there is no guarantee past performance will be indicative of future results. In preparing this publication, TNS LLC has relied upon information supplied by its customers, publicly available information and press releases which it believes to be reliable; however, such reliability cannot be guaranteed. Investors should not rely on the information contained in this website. Rather, investors should use the information contained in this website as a starting point for doing additional independent research on the featured companies. The advertisements in this website are believed to be reliable, however, TNS LLC and its owners, affiliates, subsidiaries, officers, directors, representatives and agents disclaim any liability as to the completeness or accuracy of the information contained in any advertisement and for any omissions of materials facts from such advertisement. TNS LLC is not responsible for any claims made by the companies advertised herein, nor is TNS LLC responsible for any other promotional firm, its program or its structure.
[1] A portion of Imbruvica’s net revenue is allocated to the company’s marketing partner, Johnson & Johnson.

